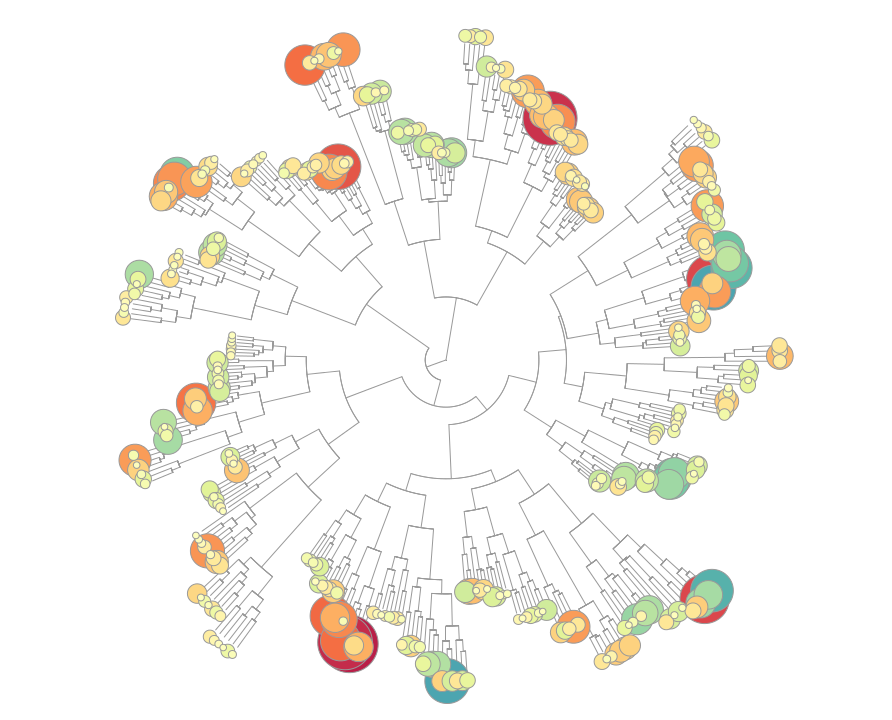
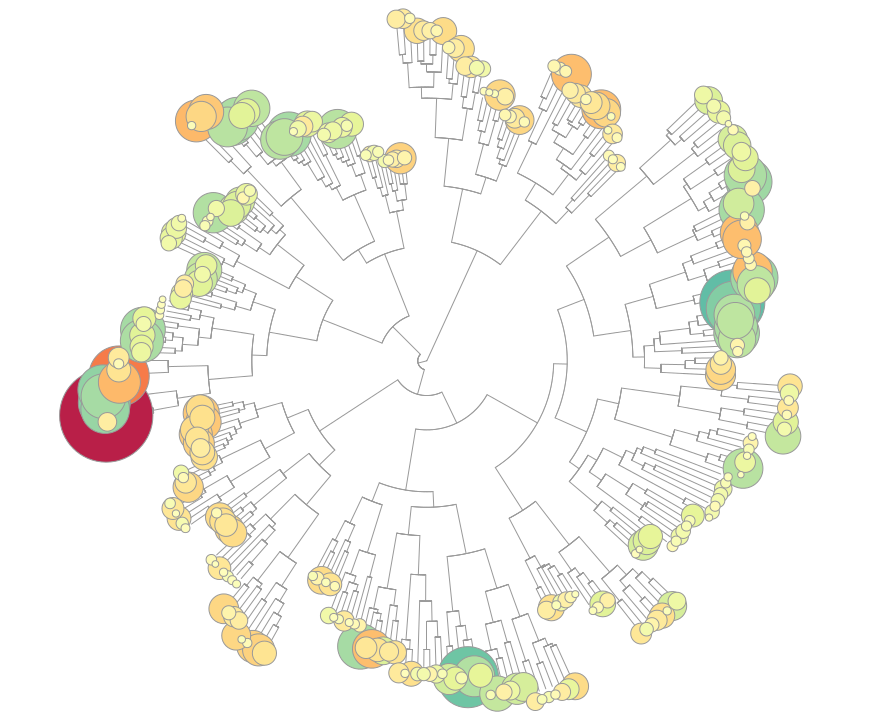
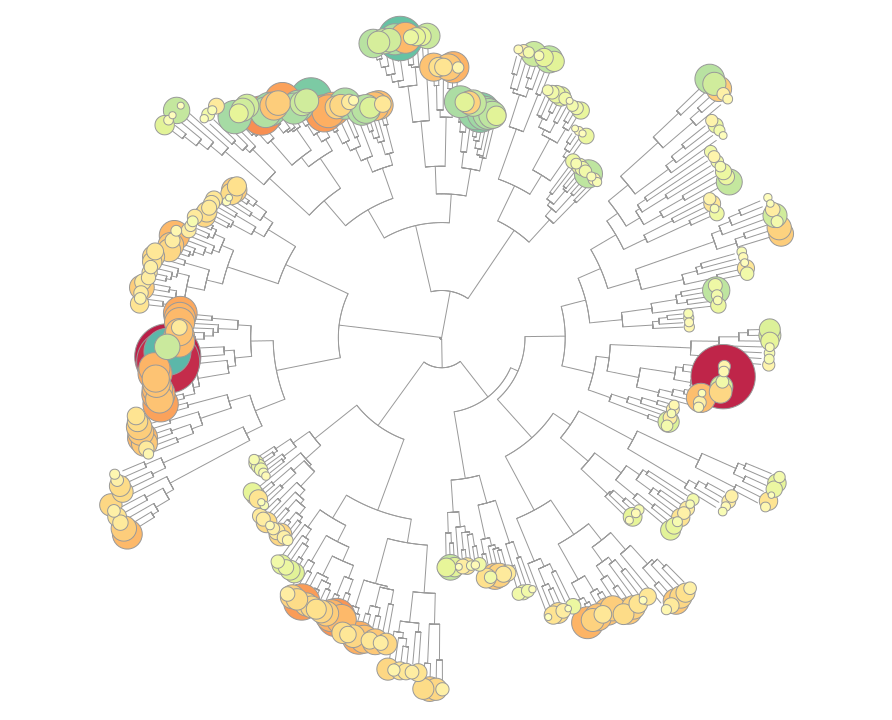
TXG-MAPr - WGCNA modules based on TG-GATEs data
LACDR - Leiden University

LACDR - Leiden University

Welcome to the TXG-MAPr app!
The TXG-MAPr app is displaying gene co-expression network analysis (WGCNA) applied to publicly available transcriptomic datasets from the TG-GATEs database. You can click on the links to go the the specified TXG-MAPr.
Introduction
Toxicogenomic data in safety testing represent a critical source to uncover underlying mechanisms of drug-induced toxicities. Co-regulated gene network approaches can organize high dimensional toxicogenomic data, while not being biased by prior gene annotations, and help in identifying novel mechanisms and regulators of toxicity. We applied weighted gene co-expression network analysis (WGCNA) on the publicly available datasets in TG-GATEs on different toxicogenomic model systems, including primary human hepatocytes (PHH), rat in vivo liver (DILI) and rat in vivo kidney (DIKI). The three datasets were independently processed with an unsigned WGCNA analysis that clustered the gene sets into functional modules, representing the bridge between individual gene variations and emergent global properties. The modules serve as a dynamic visualization of the transcriptome under experimental conditions (compounds, concentrations and time points). We developed a user friendly tool using the R Shiny package for visualization of the toxicogenomic network and analyzing the mechanism of toxicities, called the TXG-MAPr.
In the TXG-MAPr, users can analyze dose- and time-response curves, compound correlation plots and functional annotation of the WGCNA modules to derive mechanistic information of the toxicity. In addition, we included the prediction of transcription factor activities, as well as physical interactions between downstream proteins encoded by the transcriptome, which might be useful in analyzing the perturbations triggered by exposure with toxic compounds. Users can investigate module perturbation of the TG-GATEs compounds by looking at the module eigengene scores (EGS). In addition, in the upload tab it is possible to generate new module EGS using a new set of data uploaded by the user. See Sutherland et al., 2016 for details about the calculation of new module EGS.
Credits and contacts
(alphabetical order)
Callegaro Giulia1,# (g.callegaro@lacdr.leidenuniv.nl), den Hollander Wouter1, Grosdidier Solène 2, Guney Emre3, Kunnen Steven J.1,# (s.j.kunnen@lacdr.leidenuniv.nl), Mollon Jennifer4, Piñero Gonzalez Janet3, Stevens James L.1,5, Sutherland Jeffrey J.6, Trairatphisan Panuwat7, van de Water Bob1, Webster Yue5
# Corresponding authors, Division of Drug Discovery and Safety, Leiden Academic Center for Drug Research, Leiden University, Einsteinweg 55, 2333 CC, Leiden, The Netherlands
1 Leiden University, Leiden, The Netherlands
2 Erasmus MC, Rotterdam, The Netherlands
3 Hospital del Mar Research Institute (IMIM), Pompeu Fabra University (UPF), Barcelona, Spain
4 AbbVie Deutschland GmbH & Co KG, Ludwigshafen, Germany
5 Lilly Research Laboratories, Eli Lilly and Company, Indianapolis, IN, United States of America
6 Indiana Biosciences Research Institute, Indianapolis, IN, United States of America
7 Heidelberg University, Heidelberg, Germany